Bioburden Testing Market Trends: Reshaping Safety in 2026
The landscape of microbial quality control is undergoing a seismic shift. As we navigate through 2026, the demand for higher precision and faster results is no longer a luxury—it is a regulatory and operational necessity. Manufacturers are moving away from reactive testing toward proactive contamination control, fundamentally altering how products are brought to market.
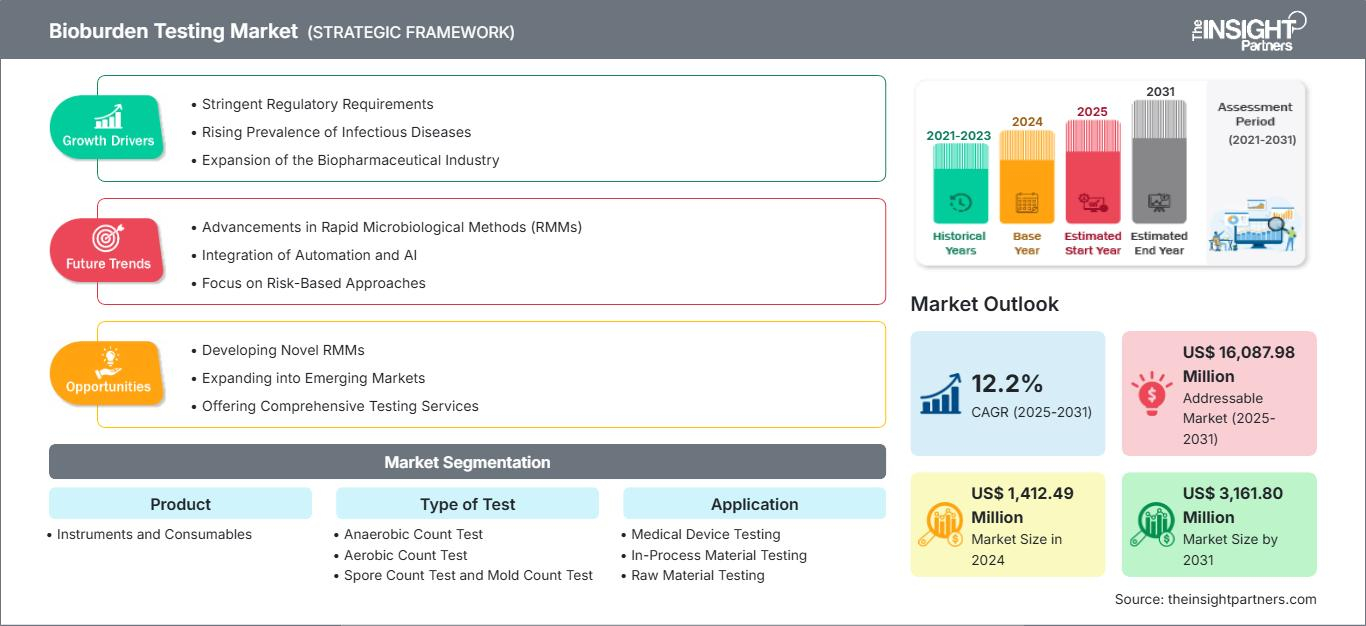
The Bioburden Testing Market size is expected to reach US$ 3,162 million by 2031. The market is anticipated to register a CAGR of 12.2% during 2025–2031. This trajectory is fueled by a convergence of technological breakthroughs and an uncompromising focus on patient safety.
Download Sample Report - https://www.theinsightpartners.com/sample/TIPRE00003000
Dominant Bioburden Testing Market Trends
One of the most significant Bioburden Testing Market Trends is the rapid adoption of Rapid Microbiological Methods (RMMs). Traditional culture based testing, which often requires 5 to 7 days for incubation, is being challenged by technologies like ATP bioluminescence and solid phase cytometry. These methods can deliver results in less than 24 hours, allowing biopharmaceutical companies to release batches faster and reduce warehouse holding costs.
Another defining trend is the integration of Automation and Robotics. Laboratories are increasingly deploying automated colony counters and liquid handling robots to eliminate human error—the leading cause of secondary contamination. By standardizing the "pulling and plating" process, manufacturers achieve higher reproducibility and data integrity, which are critical during stringent regulatory audits.
The rise of Personalized Medicine and Cell Therapies is also steering the market. Unlike traditional mass produced drugs, these therapies have short shelf lives and require immediate bioburden results. This has necessitated a shift toward "at line" testing, where microbial monitoring happens directly on the production floor rather than in a remote centralized lab.
Key Market Drivers
- Heightened Regulatory Scrutiny: Agencies like the FDA and EMA are emphasizing "Quality by Design," requiring robust bioburden data at every stage of the manufacturing process, from raw materials to finished products.
- Expansion of Outsourcing: To keep up with these Bioburden Testing Market Trends, many firms are outsourcing to specialized Contract Manufacturing Organizations (CMOs). These partners offer the high tech infrastructure needed for advanced microbial detection without the capital expenditure.
- Focus on Sustainability: There is a growing trend toward "green" microbiology, utilizing smart consumables that reduce plastic waste and energy efficient incubation systems.
Top Players Leading the Innovation
The following companies are at the forefront of these Bioburden Testing Market Trends, providing the instruments and consumables necessary for modern sterility assurance:
- Thermo Fisher Scientific Inc.
- Merck KGaA
- bioMérieux SA
- BD (Becton, Dickinson and Company)
- QIAGEN
- SGS SA
- Pacific BioLabs
- Nelson Laboratories, LLC
- Charles River Laboratories
- STERIS
Future Outlook
As we look toward 2031, the Bioburden Testing Market will likely be defined by the "Digital Lab" concept. Artificial Intelligence is already being used to predict contamination risks by analyzing environmental monitoring trends. This move toward predictive microbiology will ensure that the 12.2% CAGR is not just a reflection of volume, but a testament to the increasing value of safety data in the modern industrial world.
Related Report :
· Pharmaceutical Quality Control Market Overview, Growth, Trends, Research Report (2021-2031)
Contact Information -
Email: sales@theinsightpartners.com
Phone: +1-646-491-9876
Also Available in :
Korean German Japanese French Chinese Italian Spanish
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Jogos
- Gardening
- Health
- Início
- Literature
- Music
- Networking
- Outro
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness

